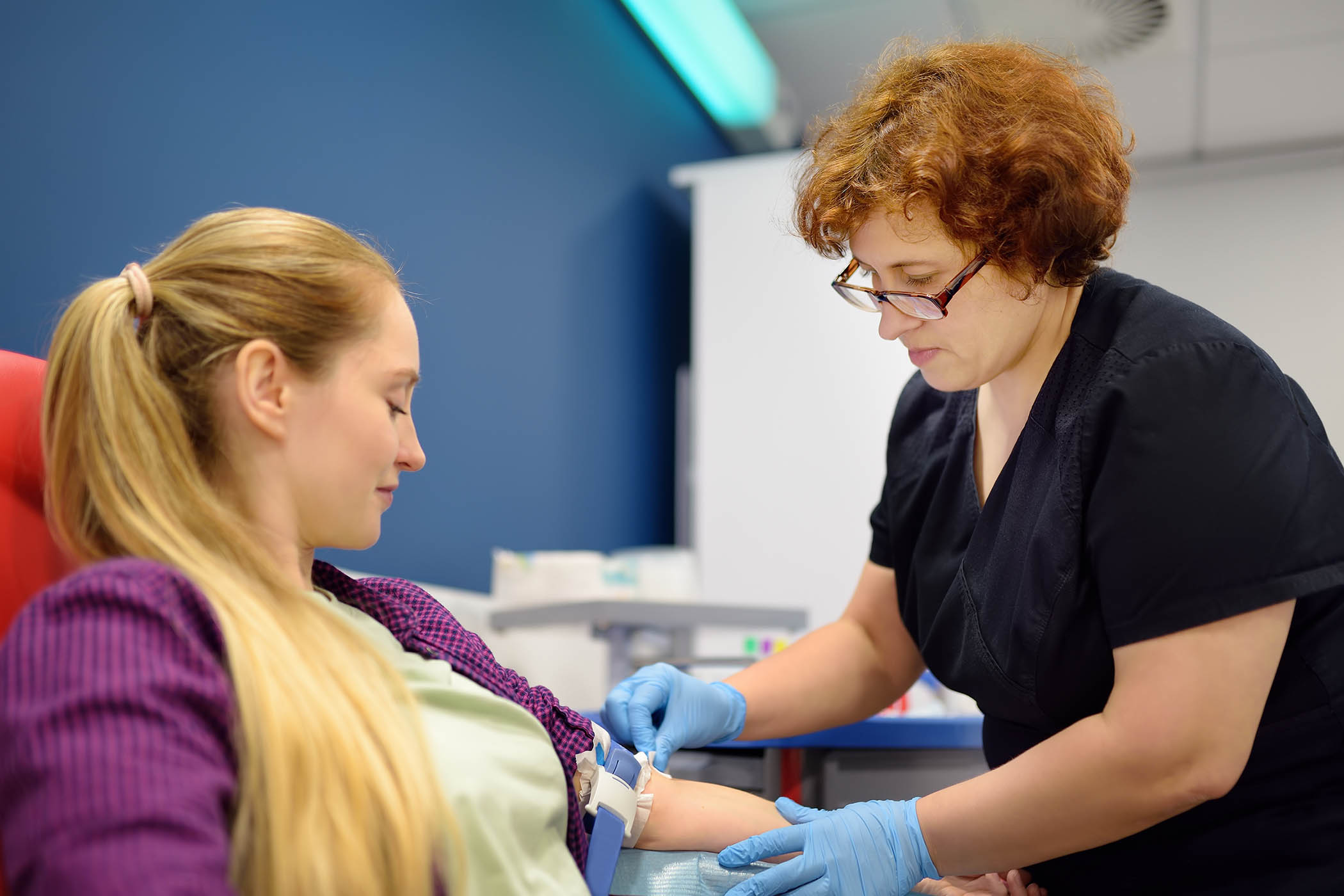
Markus Klementz is about to take his first step in seven years. His forehead puckers as he focuses into the distance. On a computer screen, his brainwaves spike and tighten with every clench of his jaw. The 39-year-old’s last steps were into a pair of skis at a resort in Chile in July 2018. Further down the slope, he tried to swerve around a slower group. Instead, he sped off a ridge and landed, back first, on a rock, ruining three of his vertebrae.
He feels lucky. “There was a rescue team that saw the accident and was there, like, five seconds later,” he says. “I [immediately] noticed that I couldn’t move my legs. It was obviously insanely painful. They transported me to the helicopter, flew me to hospital and I woke up two days later.”
Today, Klementz, a Swiss pharmacist, is standing again, strapped into a harness suspended between two handrails. He has had surgery to implant a sensor into his brain, an electrode patch into his spine and a receiver to connect them inside his midriff.
A lab researcher takes away his chair, while another sits on a stool in front of him, bracing his knees.
Standing beside them is Prof Grégoire Courtine, the French neuroscientist responsible for this impending miracle. His NeuroRestore clinic here in Lausanne in Switzerland is at the forefront of efforts to cure spinal injuries. Their newest patient is about to test their latest technology, and his own mental strength.
Klementz’s brainwaves dance along the screen, and a researcher looks up.
“When you’re ready, Markus.”
***
The oldest medical record detailing spinal injuries is an Egyptian papyrus dated to circa 1600BC. It lists the case of a tetraplegic whose dislocated vertebrae meant he had no use of his arms, legs or bladder. It was, the hieroglyphs say, “an ailment not to be treated”.
For at least 5,000 years, that was axiomatic. Physicians classed paralysis with other untreatable diseases, such as leprosy or cancer. When Sidney Farber decided in 1947 to try to treat children with leukaemia using a chemotherapy drug, his colleagues were mystified. The only thing doctors could do, they thought, was to make the children comfortable.
Newsletters
Choose the newsletters you want to receive
View more
For information about how The Observer protects your data, read our Privacy Policy
Cancer research has made astonishing progress since then, but work on paralysis has not kept pace. The number of cases has dropped significantly thanks to polio vaccines, but a spinal cord injury remains a sudden, life-shattering shock to the 4,400 people in the UK each year who suffer one, many from incidents as innocuous as falling over. Around 15 million people worldwide have suffered some sort of injury that has broken the link between brain and body.
The team at NeuroRestore are aiming to create algorithms that translate patients’ brainwaves into actions
Courtine is the embodiment of hope for them. The 50-year-old and his research partner, Prof Jocelyne Bloch, a neurosurgeon, were the first to find a way to restore that connection, using a “digital bridge” that Klementz is testing today.
“It’s hard to overstate how significant these breakthroughs are,” says Tara Stewart, the chair of Spinal Research, a UK charity, who was paralysed from the chest down when she was thrown from her horse in 2014. Like Farber’s cancer colleagues, doctors have had little to offer the newly injured. “As soon as you’re medically stable, you’re put out into the community and they say: ‘Good luck, deal with your life in a wheelchair – you’ll never walk again, you’ll never recover any function, you’ll never get any better. Deal with it. Move on.’ As a person in a chair, I can’t even begin to put into words how exciting and hopeful this is.”
Courtine’s quest began at the bottom of a rock climbing wall. The physics undergraduate fell into conversation with a professor of neuroscience, who explained to him how the brain directed his muscles to climb the wall. Inspired, he switched to neuroscience.
Courtine pursued his research at UCLA in Los Angeles where he met Christopher Reeve, the Superman actor paralysed after a riding accident. He was starstruck: the only action figure he’d ever owned was of Reeve.
“The vision of him fully paralysed was very intense for me,” Courtine says. He had found his mission. “I can’t stand the idea of people who cannot move.”
The first step on the path to unlocking a cure for paralysis was taken by another climber, Thomas Graham Brown, a Scottish mountaineer and physiologist who used electricity to stimulate the spines of paralysed cats. He demonstrated that the jolts did not simply induce random spasms; instead, the cats’ leg muscles moved as they would if they were walking.
The narrative has changed. People who are paralysed can stand, can walk
The narrative has changed. People who are paralysed can stand, can walk
Take a human and strip away bones, muscles, skin – everything except the neurons that we use to think and act, and you are left with the central nervous system. The spinal column’s motor neurons are no less complex than many of those they connect to in the brain. Courtine refers to it as “the spinal brain”. It knows what to do; it merely needs to be told when to do it.
***
Markus Klementz is being calibrated. “We’re trying to find out what are the best parameters to create the algorithm to understand that he’s attempting a right or a left movement,” says Valeria Spagnolo, a doctoral student working at NeuroRestore. She is controlling how much power is being sent to electrodes attached to muscles on Klementz’s legs, a feeling he describes as like “touching a light electric fence”.
She increases the power until his left knee flexes. “Left,” she calls, and watches his foot as it taps and slides a fraction. “We need more steam.”
***
Courtine began with rats. In May 2012, he and other researchers at the École polytechnique fédérale de Lausanne (EPFL) published a paper in Science showing that rats whose hind legs were paralysed by a partially severed spinal cord were able to walk again on their own. Some could even run.
The rats were strapped into harnesses, a short stumble away from a chocolate treat. Their brains and spinal cords were stimulated with electricity and they were given injections to encourage cell growth. After about five or six weeks of chocolate-chasing, they could walk unaided. Their nervous systems had remapped somehow, bypassing the injury by using the remaining neural connections in the spinal column. If the researchers could replicate this in humans, it would be transformative.
After news of this discovery raced around the world, Courtine felt sick. “The pressure was too high,” he said. “People started saying it will never work in humans.”
Some 4,400 people in the UK suffer a spinal injury every year
For two years he struggled to get funding, but he also found Bloch, Switzerland’s first female neurosurgeon. Courtine and his growing team in Lausanne worked on a spinal implant: 16 electrodes to stimulate the spinal column, amplifying signals from the brain. David M’zee, one of the first patients, managed to learn to walk again with crutches.
They also designed the digital bridge, announcing in 2016 they had created the first ever brain-spine interface for monkeys. Slowly, Courtine’s work gathered acceptance among the scientific community and in 2019 he was made a Rolex laureate by the Swiss watchmaker, which gave him a funding boost; Rolex continues to support his work now through its Perpetual Planet initiative.
By 2023, Courtine’s vision was realised. The digital bridge was proven to work in humans, when Dutchman Gert-Jan Oskam had two brain implants, one for each leg, which he could use to control the impulses to his legs. “The narrative has changed,” Courtine said. “We can never claim it will cure paralysis, but people who are paralysed can stand, can walk. It changes their perspective on life.”
***
“Right,” calls Valeria Spagnolo. She sends a signal to stimulate Klementz’s muscles and his right leg rises, jerks forward, then drops. He adjusts his grip on the handrail, ready for the next step. “Left.”
The computer’s AI model, BrainGPT, is learning what signals his brain implant is detecting when he tries to walk, and linking the signals with the electrical pulses sent by Spagnolo. The aim is to create an algorithm translating his brainwaves into actions.
Fifteen effortful paces later, Klementz sits down again. It’s hard for him to encapsulate the sensation of trying to move, something he stopped being able to feel seven years ago. Does he conjure a mental image of his leg, or an intention to move forwards? How would any of us describe what it’s like to do something as automatic as walking?
“I’m just trying to lift the foot,” he says. His right leg is more affected by the injury so when something is happening he gets more feedback, more feeling, from his left. “The intention, how the model is trained, is trying to keep the foot or the leg in the air. And once I release that intention, then the stimulation drops off. Basically you try not to think of what you thought of before.”
If all goes well, then by April he will have regained strength in his legs and the model will be able to decode the nuances of his brain to make walking more natural.
It will be hard work, with possibly life-changing results. But Klementz’s eyes are not flashing with excitement at these first steps. Why? He is worried about failure: there is a chance that he might not be able to train the AI. “There’s always hope that this will bring some benefit,” he says. But as a pharmacist he is also curious about the science: he hopes the research will benefit others at least as much as him.
Courtine interjects.
“We don’t want to sell dreams here,” he says. “It’s not a cure. We call people like Markus test pilots, because they are giving us feedback and allow us to develop the technology.” Their first patient, in 2016, was disappointed because he had imagined he would walk away from hospital. Courtine’s patients will still need their wheelchairs, he says. “The expectation was wrong.”
Tetraplegics and paraplegics face other problems that some see as more important than walking again. Paralysis also affects the bladder, the bowels, the sexual organs. Sitting or lying in one place creates skin sores, and damage to the nervous system can create neuropathic pain, a burning or numbness. Low blood pressure and hypotension, the feeling of lightheadedness that able-bodied people sometimes get when they stand up, can make it impossible for some to even sit upright.
As a person in a chair, I can’t put into words how exciting and hopeful this is
As a person in a chair, I can’t put into words how exciting and hopeful this is
The NeuroRestore lab and its commercial counterpart, Onward, are among research teams looking at these issues. Onward’s Arc-Ex uses electrodes attached to the skin to help people regain some use of their hands. The device has been approved in the US and Europe, and is waiting for clearance from the MHRA, the UK regulator. Another implantable prototype medical device, Arc-Im, which aims to relieve low blood pressure issues, is also in clinical trial.
These types of neuromodulation – attempts to reawaken neural pathways that were not affected by injury – are the first stage in finding a cure for spinal injuries, according to Tara Stewart of Spinal Research.
The digital bridge being tested by Klementz is the most advanced of the neuroprosthetics, enabled by rapid advances in brain interface technology. The device that Bloch implants in her patients’ skulls is less invasive than Elon Musk’s Neuralink, which threads fine platinum-iridium needles inside the cortex to detect neuron activity. Courtine and Bloch are sceptical about the ethics of these devices, since the needles can corrode. Still, competition among brain-computer interface companies is driving advances. In August, researchers at Stanford University were able to decode the inner speech of patients unable to use their mouths.
The solution that tetraplegics would prefer is finding a way to let the body repair itself, either using drugs derived from gene therapy or electrical stimulation. Spinal Research is funding a trial looking at whether the spinal cord can be encouraged to find new neural pathways if a patient intermittently breathes in low levels of oxygen, similar to altitude training.
“Scar tissue [around a spinal injury] makes it very difficult for neurons to grow back,” Stewart says. An enzyme called chondroitinase can remove scar tissue; researchers are studying ways to control it.
Perhaps the ultimate path to a cure would be a way to generate new neurons, something that stem cell researchers are considering.
Charities such as Spinal Research have struggled for funding in the past. “Spinal cord injury has always been seen as incurable,” Stewart says. “People don’t want to give their hard-earned money to something that feels hopeless.” Courtine’s research proves otherwise.
“I get really cross with consultants who tell people there’s no hope,” Stewart says. “If you take away hope from people, they give up. And it’s not true. You have to temper the fact there is hope with the kind of hope it is and how long it will take.”
She raises her hands. “Another 10% function in my right hand would transform my life because I’d be able to write. I’d be able to type. I’d be able to wear trousers with buttons, which is currently impossible because I can’t do them up.”
Markus Klementz has a different ambition. “If you’re at an event where everyone is standing, and you’re the guy in the wheelchair, it’s a bit annoying. You have to look up all the time. Being able to stand and have a conversation eye to eye...” He looks across at me, and smiles.
Melanie Reid’s view
For those of us with a long-term spinal injury, Grégoire Courtine has rock star status. His “digital bridge”, imperfect though it still is, is the best hope of progress towards a cure in our lifetimes.
Understandably, those of us who live with the 24-hour discomfort and grind of paralysis get cynical. Science takes a long time. Breakthroughs are rarely as billed. Fifteen years ago, when my accident happened, I remember being naively cheered to hear of developments that in five years’ time might transform spinal medicine. Might indeed.
Those five years came and went, as did subsequent five-year timescales. Humans need hope, but we also learn to protect ourselves against disappointment. I remember Courtine’s rats in 2012, because Sky News sent a cameraman to my home and asked me to enthuse. I tried to balance their expectations, tried to explain how all-encompassing a spinal injury is, how far from a cure this was.
Every bit of research is hugely welcome and what Courtine does is precious. During lockdown, I participated in a global trial of his Onward Arc-Ex device. Three or four days a week, for five months, the electrical messages buzzed across the break in my neck. With the help of intensive physiotherapy, I improved some strength and function in my more damaged hand, some of which has lasted. That device comes to market soon.
Spinal injury is a world of small gains and zero miracles. Science inches forwards. We crave the fantasy of the complete cure – something to make our movements smooth again, restore power, continence and normal sensation, remove the chronic neuropathic pain and spasm – but believe me, in its absence we will take every crumb of progress on offer.
Melanie Reid, a columnist for The Observer, was paralysed in 2010 in a riding accident
Photographs by Rolex/Gabriel Monnet